USP has recently announced the postponement of the official dates for revised and new general chapters 795, 797, 800 and 825 until further notice. This provides some much-needed breathing room for facilities that have been under pressure to update their pharmacy spaces by the original December 1, 2019 deadline. (Go ahead—exhale.) While delayed, change is still coming, so stay on track by keeping these things you need to know in mind:
Date Postponed for USP Pharmacy Standards: What You Need to Know
- Why the delay? Several pharmacy associations as well as the American Hospital Association have urged the delay, citing concerns with capital costs and tight schedules to become compliant as well as operational “concerns with beyond-use dates, repackaging and temperature metrics”. At this point, the appeals under consideration are not related to facility design and engineering controls, so if you have a project underway, press on.
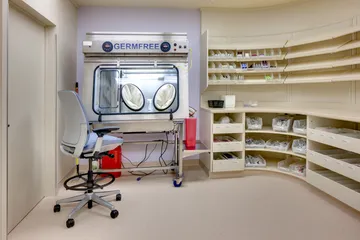
- USP 800 – Still official but only “informational” as of December 1, 2019. There are no planned changes to this chapter, so get prepared now by making a plan for compliance to create a safer environment for your staff and patients.
If you aren’t familiar with USP 800, take a look at our Insight from 2018 – Your Pharmacy Resolution: A Safer 2018 with USP 800. (Then come right back.) - Proposed revisions may already be woven into your state’s regulations. If your Board of Pharmacy has recently made revisions, they may have already incorporated some of the revised portions of USP 795 and 797. Your Board of Pharmacy may also elect to enforce USP 800—even though it’s only informational according to USP. Keep an eye out for updates to your state regulations.
- The initial time crunch has benefits! While stressful for those on the forefront, the initial deadline has proven valuable by yielding lessons learned from which you can benefit. Find a trusted advisor. (Coughs and shamelessly points to Kahler Slater contact information.) We’ve racked up quite a bit of experience—assisting over 25 clients with their pharmacy updates in USP compounding requirements over the last 2 years—and we look forward to applying that expertise to help you make your transition to compliance!

